 What kind of evidence of a disease would survive after 12,000 years? As a cause of death among animals, disease is probably a much more significant factor than predation, especially if they are under stress, though given the penchant of predators to pick out the weak and infirm, it may be difficult to distinguish (Shipman, 1981). A disease that could kill 3 sloths at once would have to have been dangerous and fast-acting, but can you prove it from fossils?
What kind of evidence of a disease would survive after 12,000 years? As a cause of death among animals, disease is probably a much more significant factor than predation, especially if they are under stress, though given the penchant of predators to pick out the weak and infirm, it may be difficult to distinguish (Shipman, 1981). A disease that could kill 3 sloths at once would have to have been dangerous and fast-acting, but can you prove it from fossils?
A hypervirulent disease has been suggested as a possible cause of the Pleistocene mass-extinction (MacPhee and Marx, 1997). The theory is humans or their dogs (or their fleas) brought a pathogen with them when they arrived in the New World, and while they were immune, the New World fauna was not, and large mammals died in unprecedented numbers–a dress rehearsal, if you will, for the devastation wreaked by measles, small pox, etc. when those pathogens arrived with Europeans 12,000 years later. But small pox didn’t exterminate dozens of species.
As a rule, disease-causing microbes adapt to a limited number of hosts with a life-style and genes that make them susceptible. Diseases that can infect widely disparate species are rare. MacPhee and Marx offer rabies and Leptospira as models for the plague microbe, though neither has the requisite characteristics today (e.g. aerosol transmission, range of hosts, etc.). You also need to assume some different social systems among the infected animals, i.e. more direct contact to maintain the plague. Of course, you don’t need to assume a super bug killed the Tarkio Valley Trio–a bad sloth-cold could have had the same effect if stress had left them vulnerable.
Proving the sloths died of a fast-acting acute disease is a challenge. Such diseases generally act on soft tissues, rarely do they leave an imprint on bone (Manchester, 1987). A long-term chronic disease has a better chance of leaving fossil evidence, but arthritis, or another such disease couldn’t kill three sloths at once. Does that just leave us with an interesting theory and no way to prove it? Maybe not.
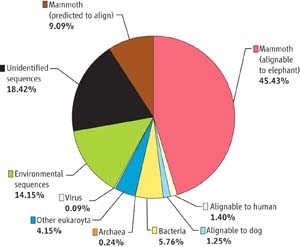 The ability to detect disease directly in fossils took a giant leap forward in 2005 when Hendrik Poinar, in the Ancient DNA Center at McMaster University in Hamilton, Ontario, and an international team, succeeded in sequencing 28 million base pairs (bp) of DNA from a well-preserved Siberian wooly mammoth jaw (Poinar et al., 2006). In a field where getting a few hundred bp of DNA is viewed as a significant achievement, they recovered 13 million bp of the mammoth’s genome. The remaining sequences were traced to a wide range of internal viruses and bacteria, a number of soil organisms, and even the mammoth’s last meal. Poinar’s team used a database of DNA sequences on file at the National Center for Biotechnology Information (NCBI), and software they developed, to identify the DNA sequences down to the species level in many cases.
The ability to detect disease directly in fossils took a giant leap forward in 2005 when Hendrik Poinar, in the Ancient DNA Center at McMaster University in Hamilton, Ontario, and an international team, succeeded in sequencing 28 million base pairs (bp) of DNA from a well-preserved Siberian wooly mammoth jaw (Poinar et al., 2006). In a field where getting a few hundred bp of DNA is viewed as a significant achievement, they recovered 13 million bp of the mammoth’s genome. The remaining sequences were traced to a wide range of internal viruses and bacteria, a number of soil organisms, and even the mammoth’s last meal. Poinar’s team used a database of DNA sequences on file at the National Center for Biotechnology Information (NCBI), and software they developed, to identify the DNA sequences down to the species level in many cases.
Sequencing DNA from our fossils is a long shot. The mammoth was preserved under ideal conditions. Iowa’s wet temperate climate makes finding DNA extremely unlikely. However, the NSF wants us to try and Poinar has agreed to help. Who knows what we’ll discover along the way–hopefully proof we have a sloth family, and maybe entire internal and external ecosystems in those fragile strands . . . . Dave
References
Poinar, HN, Schwarz, C, Qi J, Shapiro, B, MacPhee, RDE, Buigues, B, Tikhonov, A, Huson, DH, Tomsho, LP, Auch, A, Rampp, M, Miller, W, Schuster, SC. 2006. Metagenomics to Paleogenomics: large scale sequencing of Mammoth DNA. Science 311(5759): 392-294.
http://www.sciencemag.org/cgi/content/full/1123360?DC1#REF14
Rowe, B. 2005. Re disease and mass extinction The sneeze heard ’round the world: disease and the great Pleistocene extinction. Abstracts, Society for American Archaeology 70th Annual Meeting, March 30-April 3, 2005, Salt Lake City. P. 254.
MacPhee, R.D.E. and Marx, P.A. 1997. The 40,000 year plague: humans, hyperdisease and first-contact extinctions. In Natural Change and Human Impact in Madagascar. S. Goodman and B. Patterson (eds.) Smithsonian Institution Press.
Manchester, K. 1987. Skeletal evidence for health and disease. In Death, decay and reconstruction: Approaches to archaeology and forensic science. A. Boddington, AN Garland and RC Janaway (eds.) Manchester University Press.
National Center for Biotechnology Information (NCBI) http://www.ncbi.nlm.nih.gov/Taxonomy/taxonomyhome.html
Shipman, P. 1981. Life History of a Fossil: An Introduction to Taphonomy and Paleoecology, Harvard University Press.

I’m reminded of evidence of tuberculosis that was either found in museum specimens (osteological) of modern elephants or in the extinct mammoths and/or mastodons . I really wish I could remember the exact source or citation, but such an occurrence does increase the potential of disease occurring in the extinct sloths.
A better source to inquire about this would be Bruce Rothschild – although he tends to believe sloths show a number of pathologies which more than anything are just sloths being sloths!
If the sloth fossils have been buried in wet clay for 12,000 years is there much chance you’ll be able to recover any DNA? I understand that’s about as bad as it gets, right? If sitting frozen in ice is best, wouldn’t sitting DRY in a cave be your only other hope?
Rob, thanks for the terrific tip. Rothschild published a paper on the subject two years ago. see http://news.nationalgeographic.com/news/2006/10/061003-mastodons.html He believes it only weakened the animals and didn’t kill them outright but that virtually every one was infected. Add tuberculosis to the list of pathogens that infects many species. Per NCBI, Mycobacterium tuberculosis and M. tuberculosis-like organisms have been identified in a wide range of species, including primates, elephants, ungulates, carnivores, marine mammals and parrots. However, it’s normally limited to captive settings and does not occur naturally in free-living populations (that’s the social system barrier MacPhee refers to). See http://www.ncbi.nlm.nih.gov/pubmed/11288517 Low virulence and poor transmission knock M. tuberculosis out of contention for the super bug, at least in its current form, but Rothschild would say keep your mind open for it still being a contributing factor. Thanks again.
Travis, You’re right. We would have preferred cool and dry conditions for preservation. It would have been better still if we had found the sloths embedded in a glacier, but wet anoxic conditions similar to ours have yielded DNA too. (See Hagelberg and Clegg, 1991, Lawlor et al., 1991, Paabo et al., 1988) Even at a single site conditions can vary a lot and produce different results with respect to DNA recovery. (See Stone and Stoneking, 1999; Hagelberg, E, Clegg JB. 1991.) The other important point is the hydroxyapatite mineral structure of bone offers some protection to the biomolecules inside, including DNA–the better the bone preservation, the better the chances for DNA survival (Hagelberg et al. 1989). The first look at our bone says the histology is excellent. So there are good reasons to be hopeful.
Hagelberg, E, Sykes, B, Hedges, R. 1989. Ancient bone DNA amplified. Nature 342: 485.
Hagelberg, E, Clegg JB. 1991. Isolation and characterization of DNA from archaeological bone. Proceedings of the Royal Society of London. Biological Science 244: 45-50.
Lawlor, DA, Dickel, CD, Hauswirth, WW, Parham, P. 1991. Ancient HLA genes from 7,500-year-old archaeological remains. Nature: 349: 785-788.
Paabo, S., Gifford, JA, Wilson, AC. 1988. Mitochondrial DNA sequences from a 7000-year-old brain. Nucleic Acids Research 16: 9775-9787.
Stone, AC, Stoneking, M. 1999. Analysis of ancient DNA from a prehistoric Amerindian cemetery. Philosophical Transactions. Royal Society of London, B. Biological Science: 354(1379): 153-159.